A regenerative approach to soft tissue reconstruction
Soft tissue reconstruction requires more than restoring volume, it requires rebuilding living, functional tissue.
AxCell’s approach is based on a structured, implantable scaffold that supports cell migration, tissue integration, and vascularization. Rather than simply filling space or stimulating tissue externally, our technology is designed to guide the formation of new, healthy tissue from within.

Early tissue integration following subcutaneous implantation
A novel implantable biomaterial
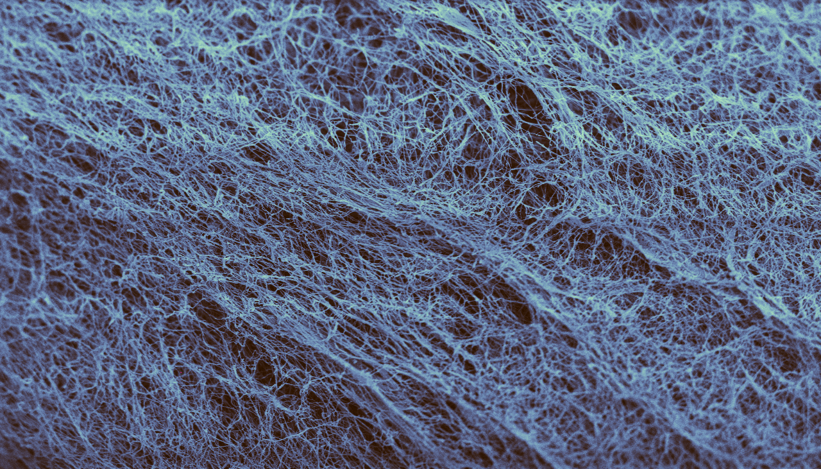
At the core of AxCell’s technology is a biomaterial derived from
Bacterial Cellulose.
This material forms a highly structured, ECM-like matrix that enables cell colonization and tissue ingrowth, while maintaining mechanical stability and long-term integrity. Unlike traditional biological matrices, it is not derived from animal or human tissue, avoiding many of the limitations associated with those materials.
Its use as an implantable material is enabled by a proprietary purification process (patent pending) that preserves its properties while ensuring high biocompatibility.
Designed as a new class of products
AxCell’s technology is designed to function fundamentally differently from existing solutions. Instead of acting as a temporary filler or an inert implant, it provides a stable, colonizable scaffold that integrates with host tissue and supports long-term reconstruction.
This approach enables a shift from volume replacement to true tissue regeneration.
Regenerative Dermal Injectable
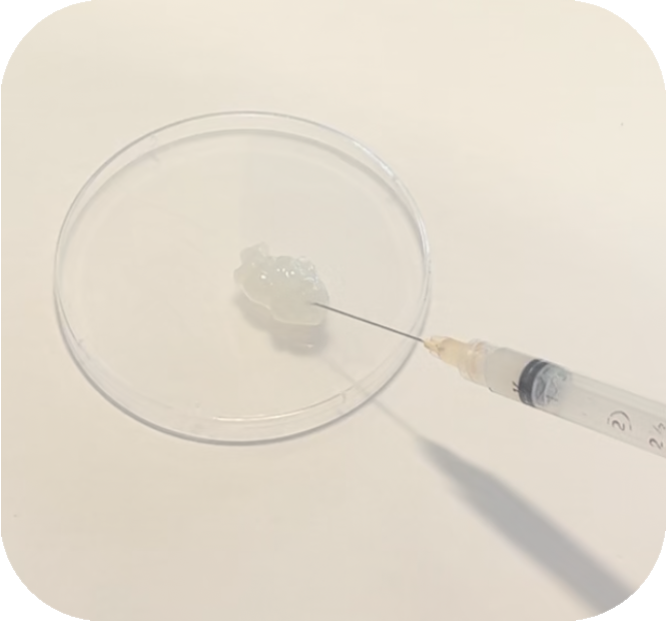
Designed to restore volume through a structured, colonizable scaffold that integrates with host tissue and supports the formation of new, vascularized tissue.
Unlike particulate-based approaches, this mechanism relies on guided tissue integration rather than inflammatory response, enabling a more predictable and controlled biological outcome.
ENT Soft Tissue Implants (in dev.)

Designed for structural soft tissue repair using a stable, biocompatible scaffold that supports tissue integration in ENT and cartilage reconstruction.
Unlike inert or inflammatory-driven implants, it enables controlled tissue ingrowth within a defined structure, supporting predictable integration and long-term reconstruction of complex tissues.
Publications
In vitro and in vivo biocompatibility of bacterial cellulose
Girard, V. D., Chaussé, J., Borduas, M., Dubuc, É., Iorio-Morin, C., Brisebois, S., & Vermette, P. (2024). In Vitro and In Vivo Biocompatibility of Bacterial Cellulose. Journal of Biomedical Materials Research Part B: Applied Biomaterials, 112(10), e35488. https://doi.org/10.1002/jbm.b.35488
